Data Protection
The Swedish Rheumatology Quality Register (SRQ) is a national quality register with the aim of improving the treatment and follow-up of patients with rheumatic diseases. All data registered in SRQ is protected by the Data Protection Regulation (GDPR), but also the Patient Data Act (Patientdatalagen). In addition, the health confidentiality in the Public and Privacy Act (Offentlighets- och sekretesslagen) also applies to quality registers.
Here you will find the most frequently asked questions and answers regarding data protection and SRQ. If you have a question that you do not find an answer to below, please contact us.
What legal basis is there for the processing of personal data in SRQ?
In order to be allowed to process personal data, there must always be support in the Data Protection Regulation, a so-called legal basis. The legal basis for SRQ is that the register is needed to fulfill a task of general interest, as well as for research and statistics.
What is the purpose of registration in SRQ?
The Swedish Rheumatology Quality Register (SRQ) is a national quality register with the aim of continuously improving the treatment and follow-up of patients with rheumatic diseases. Patients and caregivers enter information in the register and can then in real time access the patient’s health data and make a joint decision on continued care. The register also serves as a basis for research that seeks to increase knowledge and better treatment of rheumatological diseases.
What type of data is recorded in SRQ and how is it stored?
In SRQ there is mainly structured data, with a small amount of unstructured data (free text, for example comments). Data is recorded in a database that is managed by the personal data assistant (personuppgiftsbiträdet) in a national system. No data from SRQ is stored locally with the caregivers, but the caregivers have access to their own data.
Is there a personal data assistant (external party) for the processing of personal data?
Yes, SRQ’s personal data assistant is OMDA Health Analytics AB and there is a national personal data access agreement for the processing. OMDA Health Analytics provides forms for data storage, data entry for healthcare providers and patients, as well as a platform for data analysis and data collection = system supplier, cloud service provider, technical manager, developer.
Is there a written personal data entry agreement?
Yes, a national personal data access agreement exists between CPUA Karolinska University Hospital and OMDA Health Analytics AB for data management.
Must consent be given for registration in SRQ?
National and regional quality registers may process personal data as long as the individual does not oppose it (Chapter 7, Section 2, Patient Data Act – Patientdatalagen). A prerequisite for this procedure is that the patient receives information in accordance with Chapter 8 § 6 and Chapter 7 3 § PDL.
Who is responsible for personal data in SRQ?
The local personal data controller is the reporting healthcare provider (health care provider is a state authority, county council and municipality that performs health care = public health care provider, as well as another legal person, for example, a company-owned hospital that is county council-owned, or an individual trader who provides health care = private health care provider).
The central data controller is Karolinska University Hospital.
Are any sensitive data registered in SRQ?
Yes, patient data about health status is counted as sensitive data and this is registered in SRQ.
Which kinds of personal data are processed in the register?
Name, date of birth and social security number are registered in SRQ. Address, telephone number and similar information are not registered.
What is the basis for registration of social security numbers?
Social security number is used for secure identification of the person. The entered data is important for the care and is followed by the care provider. The social security number is also used for secure login to 1177.se where the Patient’s Self Registration (PER) of health data takes place.
What categories of users have access to the personal data in the register?
Personnel at the health care provider who have been granted special authorisation can use the register and have access to registered information. Within the national organization, there are some users who have and need access to the entire SRQ (national quality coordinators and statisticians).
Can information from the register be provided to outsiders?
Yes, after approval by the ethics review board and special examination locally or nationally, data can be disclosed to researchers. Research is one of the aims of SRQ. Extradition to third countries (outside the EU/EEA) does not take place.
How does the registered person get information about the register and registration?
Before the patient gives his or her consent, oral information about SRQ is provided. There are information sheets to print out in connection with the inclusion and this information is also available on the SRQ website (both the website for patients and the website for health professionals). Further information and general information about SRQ can be found on the website.
Where does my information end up?
When you make a PER registration, your information will be stored in a separate database. Your healthcare provider can see that there is an active PER registration and can then import the information.
Healthcare providers can choose to use a decision support tool, RDS, to be able to monitor your care and health over time. This system displays information from the PER registrations you have made and that have been imported by the healthcare provider, as well as information entered into the decision support system by the healthcare provider.
Every night, the information entered into the decision support system is sent to the national quality registry, SRQ (Figure 1).
 Figure 1. The relationship between the patient, PER, SRQ, RDS, and healthcare.
Figure 1. The relationship between the patient, PER, SRQ, RDS, and healthcare.
You can read more about how your information is used in SRQ in the patient information. The patient information also contains more details about your rights as a patient.
The patient information about SRQ states that you have the right to request that your information in SRQ be deleted. This is called opt-out. For the decision support tool RDS, however, it is up to your healthcare provider to decide whether or not to use it. This means that you can use PER to register your health information for local use, even if you have requested opt-out. It is clearly visible in the decision support system if you have chosen not to have your information sent to the quality registry SRQ (Figure 2).
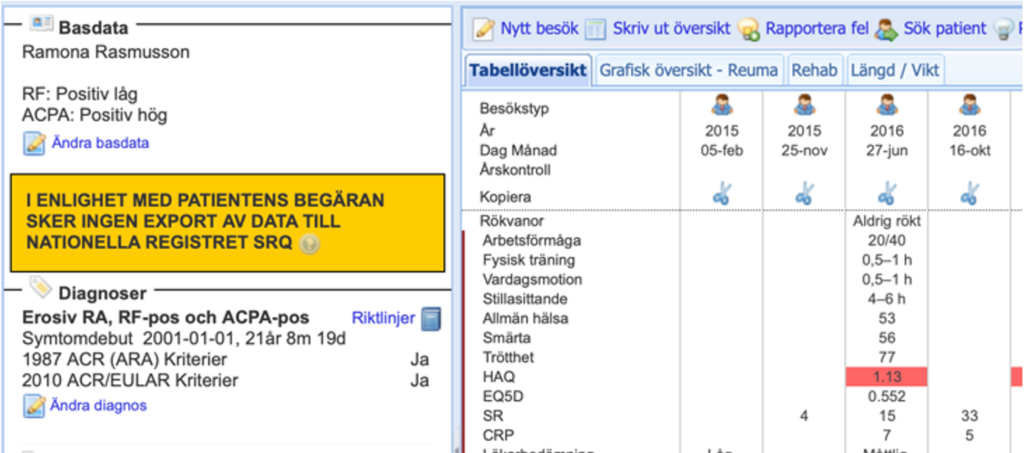 Figure 2. Screenshot of how the opt-out marking appears in RDS.
Figure 2. Screenshot of how the opt-out marking appears in RDS.
More questions
When was SRQ started?
SRQ was started on a small scale in 1995.
When will registration in SRQ cease?
Storing of data in SRQ is valid until further notice.
How many registered persons are there in SRQ?
In 2024 there were more than 135,000 registered patients in SRQ.
What data protection legislation applies to the processing of personal data in SRQ?
SRQ applies the Data Protection Regulation (Dataskyddsförordningen, GDPR) but also the Patient Data Act (Patientdatalagen).
Data Protection and srq.nu
You may choose to prevent this website from aggregating and analyzing the actions you take here. Doing so will protect your privacy, but will also prevent the owner from learning from your actions and creating a better experience for you and other users.
This page was last updated 28 november 2024.

